Few substances have captured global attention like PFAS (per- and polyfluoroalkyl substances). Found in everyday items from textiles to food packaging, these chemicals are prized for their heat and water resistance. Yet their strength is their danger. At their core lies the carbon-fluorine bond, a “molecular superglue” that prevents them from breaking down in the environment or the human body. As these "forever chemicals" accumulate, they pose a lasting challenge to public health.
From wonder to worry
PFAS’s journey began in 1938 with the accidental discovery of Teflon (PTFE). By the 1950s, these "synthetic marvels" became ubiquitous. Prized for being water-repellent, grease-resistant, and heat-stable, they evolved into the invisible workhorses of the modern world, found in everything from non-stick pans and raincoats to firefighting foams and medical implants.
However, the very durability that made PFAS a wonder of the 20th century has created one of the most urgent environmental challenges of the 21st. In nature, there is no mechanism to break the incredibly strong carbon-fluorine bond. This way, these "forever chemicals" have become ubiquitous, contaminating soil and groundwater on a global scale. Originating from industrial leaks, landfills, and firefighting foams, they migrate effortlessly through water systems to even remote polar regions. Because they do not biodegrade, they cycle endlessly through the food chain, from soil to water to blood, accumulating in the body of nearly every person on Earth.
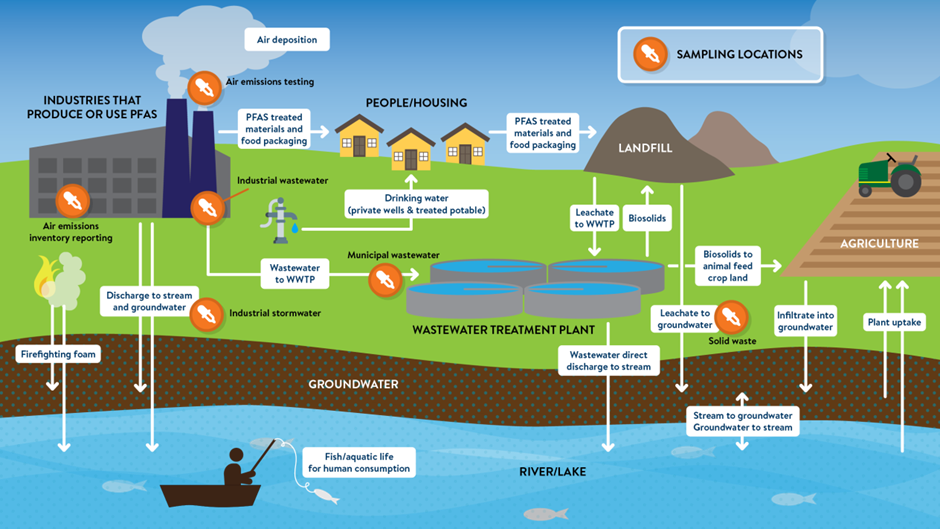
Figure 1: The PFAS-cycle (Ecopulsenow.com)
Once PFAS enter the human body, they bind to proteins in the blood and settle deep within vital tissues, particularly the liver and kidneys. This process of bioaccumulation creates a toxic reservoir that grows over time. Because the half-life of common variants like PFOS (Perfluorooctane sulfonic acid) and PFOA (Perfluorooctanoic acid) ranges from two to eight years, it would take nearly a decade for the body to eliminate just half of its burden, even if all exposure ceased today.
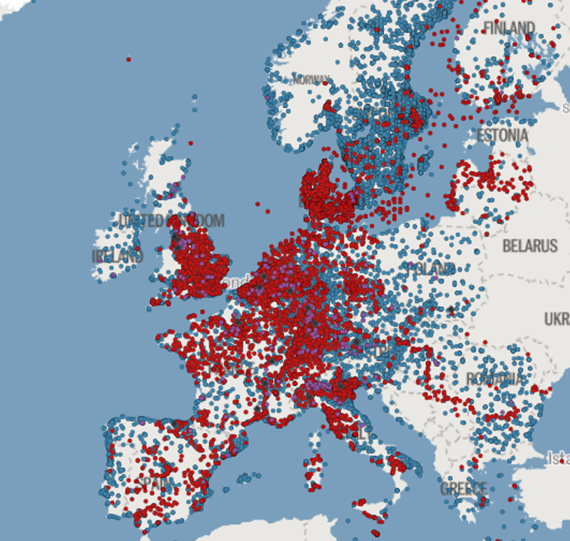

Figure 2 : Map of
Europe showing known and presumptive contamination sites
(Source: Forever Pollution Project)
Hospitals as PFAS-hotspots
While the medical community increasingly discovers the health impacts of PFAS, the clinical environment itself acts as a significant reservoir for exposure. Valued for their exceptional chemical inertness, thermal stability, and ability to repel water, lipids, and blood, PFAS have become deeply embedded in modern healthcare.
Medical goods
At the core of clinical intervention, fluoropolymers like Polytetrafluoroethylene (PTFE) and Fluorinated Ethylene Propylene (FEP) form the functional backbone of high-tech medicine. In interventional cardiology and radiology, PTFE remains the "gold standard" for lubricity. Catheter liners rely on this material to ensure guidewires pass without friction. Vascular grafts made of expanded Polytetrafluoroethylene (ePTFE) are currently considered essential for bypass surgery and dialysis access due to their specific porosity, allowing tissue ingrowth without triggering clotting. Hernia repairs frequently utilize Polyvinylidene Fluoride (PVDF) meshes for their resistance to degradation. Life-saving technologies like dialysis membranes, oxygenators, and the hydrophobic venting filters in IV lines depend on fluoropolymers to prevent protein fouling and bacterial ingress.
Surgical gowns and drapes utilize fluorinated durable water repellents to meet penetration standards. However, a significant "over-specification issue" exists: many hospitals routinely use high-PFAS protection for low-risk procedures where PFAS-free alternatives would suffice. This "just-in-case" use drives unnecessary consumption and leads to environmental contamination.
The ubiquitous nature of PFAS also extends to routine clinical consumables, such as wound care products. For example, certain catheter dressings (such as 3M’s Tegaderm) have historically relied on PFAS in their production processes. However, 3M has committed to completely phasing out PFAS from its entire product portfolio by the end of 2025, paving the way for PFAS-free wound dressings from 2026 onwards.
Pharmaceuticals
Beyond medical goods, pharmaceuticals are a significant pathway for PFAS entry into the environment via both drug formulations and their packaging. Approximately 20% of modern medicines, including widely used treatments like fluoxetine, celecoxib, and sitagliptine, contain fluorine atoms to prevent rapid metabolic breakdown. While these do not typically bioaccumulate in human tissue like legacy PFASs such as PFOA, their chemical stability ensures long-term environmental persistence.
This stability is particularly problematic with volatile anaesthetic gases like sevoflurane and desflurane. Because only a tiny fraction of these gases is metabolized by the patient (0,02% for desflurane and 5% for sevoflurane), the vast majority is vented directly into the atmosphere. Once released, these agents degrade into trifluoroacetic acid (TFA), a "forever chemical" that accumulates in global rainwater and aquatic ecosystems. Unlike PFOA, TFA is highly mobile and water-soluble; while it has lower acute toxicity, it is nearly impossible to filter, leading to its accumulation in global rainwater and aquatic ecosystems. Furthermore, pressurized metered-dose inhalers (pMDIs) often rely on PFAS-based propellants.
The environmental footprint extends to pharmaceutical packaging as well; blister packs frequently utilize Polychlorotrifluoroethylene (PCTFE) films to ensure drug stability through moisture barriers. While PCTFE is a stable polymer with low immediate toxicity to the consumer, its primary risk lies in the potential release of fluorinated precursors during manufacturing and hazardous gases during waste incineration.
Infrastructure
Finally, the hospitals infrastructure reinforces this toxic cycle. Because hospitals are high-traffic, high-hygiene environments, facility managers rely on "performance" materials. Resilient flooring is sealed with PFAS-based waxes to resist stains and scuff marks, while waiting room upholstery is treated with stain repellents to lower cleaning costs. In catering, grease-resistant packaging migrates chemicals into hot meals. Until the European ban in 2025, fire safety relied on Aqueous Film Forming Foam (AFFF), turning essential safety equipment into a latent environmental hazard.
Tightening the regulatory net
The search for safer alternatives is no longer just an ambition, REACH, the EU’s primary regulatory framework designed to protect human health and the environment, requires companies to identify and manage the risks linked to the chemicals they manufacture and market. REACH targets the reduction and ban of over 10,000 PFAS substances. While the goal is a ban on manufacture and import, the legislation, likely to enter into force in 2027, recognizes the unique complexity of healthcare.
To prevent disrupting critical care, specific time-limited derogations are in place. Applications where alternatives are currently in development may receive a 5-year transition. Implantable devices like stents, requiring rigorous re-engineering and Medical Device Regulation (MDR) approval, may be granted up to 12 years. However, it is a ticking clock for innovation. The regulations hinge on the "Essential Use" concept. Under this framework, PFAS use is only permitted if it is critical for health and no technical or economic alternatives exist. Consequently, while an ePTFE vascular graft remains defined as essential, a PFAS-coated surgical gown for a routine check-up does not.
Roadmap to PFAS-free hospitals
Before any strategic phase-out can begin, institutions must establish a PFAS inventory. This requires an audit and inquiries with suppliers where fluorinated compounds are currently utilized. By categorising these substances based on their function, hospitals can distinguish between "non-essential" uses and "currently essential" clinical applications. This diagnostic phase serves as the baseline for monitoring the transition, ensuring that no critical care pathway is disrupted during the shift toward safer alternatives.
To effectively transition toward a PFAS-free healthcare sector, hospitals must leverage their collective procurement power to dismantle the cycle of "forever chemicals." By prioritising environmental stewardship over mere price points, institutions can mandate chemical transparency in supplier contracts. This allows hospitals to prioritize PFAS-free alternatives for high-volume consumables, while strictly reserving fluoropolymers for critical applications where no clinical equivalent exists, effectively forcing a market-wide pivot toward safer medical materials. This focus should be mirrored in textile management, where a rationalized use of PPE can curb the over-specification of fluid-repellent fabrics, ensuring that chemically treated textiles are utilized only when clinically essential.
This procurement shift extends into clinical practice, where the "pharmacological footprint" can be significantly reduced through evidence-based substitution. Where clinical efficacy is equivalent, phasing out fluorinated medications can become a priority. For instance, transitioning from volatile anaesthetic gases (acting also as potent greenhouse gases) to total intravenous anaesthesia offers immediate dual benefits for patient and planetary health. Similarly, replacing pressurized metered-dose inhalers with dry-powder alternatives eliminates the reliance on fluorinated propellants.
Infrastructure improvements also offer possibilities in reducing exposure. By specifying PFAS-free furnishings and hard flooring during renovations, the acuumulation of toxic dust in patient wards can be drastically lowered. Furthermore, facilities could retrofit infrastructure with Granular Activated Carbon (GAC) or Reverse Osmosis (RO) filtration systems to capture upstream contaminants before they reach the tap. Simultaneously, facility managers must address legacy risks, particularly by ensuring AFFF is effectively phased out in favour of fluorine-free firefighting alternatives.
Implementing these transitions requires a coordinated effort across all levels of the organization. To move from the understanding of PFAS risks to a concrete operational shift, we recommend the following steps:
- Identify where PFAS are embedded in your hospital, categorising items into "non-essential" and “clinically vital".
- Demand clear roadmaps for PFAS alternatives from key suppliers, mandating the disclosure of specific fluorinated materials in new tenders.
- Bridge the gap between departments by educating clinical, procurement, and facility teams on the health risks and environmental costs of "forever chemicals."
At NZHI, we bridge the gap between sustainability goals and clinical reality. We assist hospitals to dismantle the cycle of "forever chemicals" by mapping hazardous substances within your specific processes, evaluating wastewater contamination, and identifying alternatives to legacy products. From procurement to patient care, we support healthcare institutions at every stage of the transition utilising science-based tools and hands-on expertise. Let us help you move from intent to impact.
We thank Prof. Dr. Daan Van Brusselen for his expert review and valuable contribution to this article.
References
- Briassoulis, G. Et al., 2025, Exposure to Per- and Polyfluoroalkyl Substances (PFASs) in Healthcare: Environmental and Clinical Insights, Life
- European Commission: Directorate-General for Environment, 2026, The cost of PFAS pollution for our society – Final report, https://data.europa.eu/doi/10.2779/9590509
- Van Brusselen Daan. Dagboek van een kinderarts zonder grenzen. Borgerhoff & Lamberigts 2025 (264 blz),
- Uijtewaal, A. Et al, 2025, PFAS-vrij alternatief beschikbaar voor veel PFAS-medicijnen, Pharmaceutisch weekblad
